

圆形微腔设计太大使得溶液浪费且测试过程中基底的更换、池体的密封困难。然后完成圆环型微元腔体的设计及制作,其足够小的腔体减小了溶液对椭偏测试带来的影响。但是容纳的溶液会带来沉积离子不够的问题且经过实验发现对电极ITO上会出现气泡,影响椭偏测试,所以在此基础上设计制作完成了长方形微流腔体。
展示全部 
椭偏仪在位表征电化学沉积的系统搭建(二十)- 长方形流动微腔
3.3.2长方形流动微腔
为了解决溶液微圆形腔体溶液注入困难、溶液少及反应产生气泡的问题,进一步对腔体进行改进,得到微流腔体。图3-18是微流腔体制作过程示意图,首先准备好ITO、EVA胶膜、特氟龙细管、Au/Si,把EVA胶膜切成内为25px×62.5px,外为37.5px×75px的长方形,然后从上到下依次把ITO、EVA胶膜/特氟龙细管、Au/Si叠好置于加热平台,在150℃下加热,使得EVA软化粘合池体,zui后冷却得到成品。
该池体工作电极即为Au/Si基底,上端的ITO即为对电极,溶液的进出由两边的特氟龙细管实现,通常制作完成后的微腔厚度和特氟龙细管一致,约为1mm。该微腔具有液层薄、溶化充足且流动可以消除ITO上产生的气泡等优点。
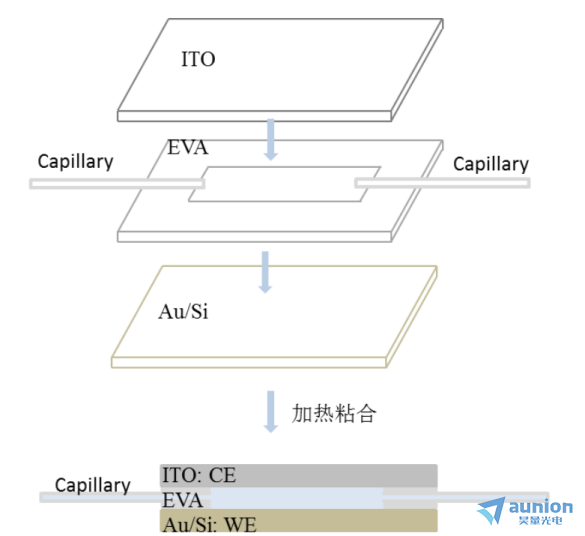
图31-18流动型微腔示意图
用该池体对不同浓度醋酸铅溶液进行测试,得到的结果如图3-19所示。可以看到有无溶液加入,测试得到的椭偏参数峰位及数值上都存在差别。但是在加不同浓度的溶液(去离子水、1M醋酸钠、1M的醋酸钠和5/10/15/20mM的醋酸铅)后得到的椭偏参数数值和趋势都一致。这和前面所述的半圆弧型电解池在不同浓度的醋酸铅溶液中椭偏测试结果一致,同样说明在醋酸铅溶液中,其浓度椭偏测试参数的影响可忽略不计。
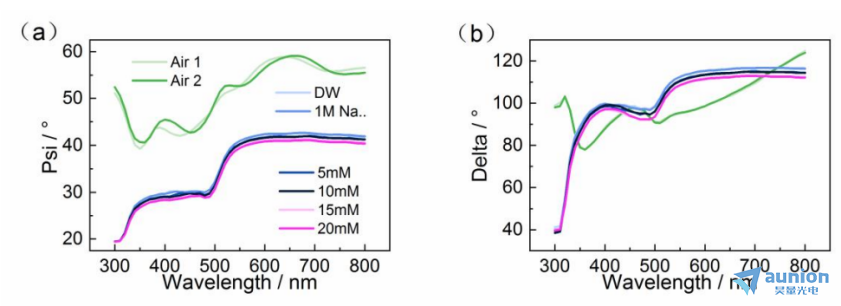
图3-19不同条件下EVA腔体椭偏测试结果(a)Psi;(b)Delta
如图3-20所示,是用该池体进行沉积薄膜的结果。电解液为0.02MCu(CH3COO)2,0.1MCH3COONa,Au/Si为工作电极,ITO为对电极,-0.4mA恒压沉积。对比沉积前后腔体图可知,用该池体可以进行沉积。与前圆环电极对比可以看到,ITO上不再有气泡存在,因为产生的气泡都被流动的溶液带走了。因溶液可以流动,故可克服圆环电极溶液少的缺点。所以后续沉积薄膜实验的椭偏仪监测选用该流动池体进行。

图3-20流动型微腔(a)沉积前(b)沉积后实物图
3.4小结
本文主要介绍了研究中实验装置的设计及测试的过程,主要包含半圆弧型器件微元腔体器件。首先设计完成半圆弧器件,实现了把沉积过程和椭偏仪测试相结合,观察窗口选用石英玻璃,理论上zui大限度减小了光的损耗。但是它的池体设计太大使得溶液浪费且测试过程中基底的更换、池体的密封困难。然后完成圆环型微元腔体的设计及制作,其足够小的腔体减小了溶液对椭偏测试带来的影响。但是容纳的溶液会带来沉积离子不够的问题且经过实验发现对电极ITO上会出现气泡,影响椭偏测试,所以在此基础上设计制作完成了长方形微流腔体。该设计成功解决了气泡和溶液少所带来的问题,故而后续实验将采用长方形微流腔体。
了解更多椭偏仪详情,请访问上海昊量光电的官方网页:
https://www.auniontech.com/three-level-56.html
更多详情请联系昊量光电/欢迎直接联系昊量光电
关于昊量光电:
上海昊量光电设备有限公司是光电产品专业代理商,产品包括各类激光器、光电调制器、光学测量设备、光学元件等,涉及应用涵盖了材料加工、光通讯、生物医疗、科学研究、国防、量子光学、生物显微、物联传感、激光制造等;可为客户提供完整的设备安装,培训,硬件开发,软件开发,系统集成等服务。
您可以通过我们昊量光电的官方网站www.auniontech.com了解更多的产品信息,或直接来电咨询4006-888-532。
相关文献:
[1] WONG H S P, FRANK D J, SOLOMON P M et al. Nanoscale cmos[J]. Proceedings of the IEEE, 1999, 87(4): 537-570.
[2] LOSURDO M, HINGERL K. ellipsometry at the nanoscale[M]. Springer Heidelberg New York Dordrecht London. 2013.
[3] DYRE J C. Universal low-temperature ac conductivity of macroscopically disordered nonmetals[J]. Physical Review B, 1993, 48(17): 12511-12526. DOI:10.1103/PhysRevB.48.12511.
[4] CHEN S, KÜHNE P, STANISHEV V et al. On the anomalous optical conductivity dISPersion of electrically conducting polymers: Ultra-wide spectral range ellipsometry combined with a Drude-Lorentz model[J]. Journal of Materials Chemistry C, 2019, 7(15): 4350-4362.
[5] 陈篮,周岩. 膜厚度测量的椭偏仪法原理分析[J]. 大学物理实验, 1999, 12(3): 10-13.
[6] ZAPIEN J A, COLLINS R W, MESSIER R. Multichannel ellipsometer for real time spectroscopy of thin film deposition from 1.5 to 6.5 eV[J]. Review of Scientific Instruments, 2000, 71(9): 3451-3460.
[7] DULTSEV F N, KOLOSOVSKY E A. Application of ellipsometry to control the plasmachemical synthesis of thin TiONx layers[J]. Advances in Condensed Matter Physics, 2015, 2015: 1-8.
[8] DULTSEV F N, KOLOSOVSKY E A. Application of ellipsometry to control the plasmachemical synthesis of thin TiONx layers[J]. Advances in Condensed Matter Physics, 2015, 2015: 1-8.
[9] YUAN M, YUAN L, HU Z et al. In Situ Spectroscopic Ellipsometry for Thermochromic CsPbI3 Phase Evolution Portfolio[J]. Journal of Physical Chemistry C, 2020, 124(14): 8008-8014.
[10] 焦杨景.椭偏仪在位表征电化学沉积的系统搭建.云南大学说是论文,2022.
[11] CANEPA M, MAIDECCHI G, TOCCAFONDI C et al. Spectroscopic ellipsometry of self assembLED monolayers: Interface effects. the case of phenyl selenide SAMs on gold[J]. Physical Chemistry Chemical Physics, 2013, 15(27): 11559-11565. DOI:10.1039/c3cp51304a.
[12] FUJIWARA H, KONDO M, MATSUDA A. Interface-layer formation in microcrystalline Si:H growth on ZnO substrates studied by real-time spectroscopic ellipsometry and infrared spectroscopy[J]. Journal of Applied Physics, 2003, 93(5): 2400-2409.
[13] FUJIWARA H, TOYOSHIMA Y, KONDO M et al. Interface-layer formation mechanism in (formula presented) thin-film growth studied by real-time spectroscopic ellipsometry and infrared spectroscopy[J]. Physical Review B - Condensed Matter and Materials Physics, 1999, 60(19): 13598-13604.
[14] LEE W K, KO J S. Kinetic model for the simulation of hen egg white lysozyme adsorption at solid/water interface[J]. Korean Journal of Chemical Engineering, 2003, 20(3): 549-553.
[15] STAMATAKI K, PAPADAKIS V, EVEREST M A et al. Monitoring adsorption and sedimentation using evanescent-wave cavity ringdown ellipsometry[J]. Applied Optics, 2013, 52(5): 1086-1093.
[16] VIEGAS D, FERNANDES E, QUEIRÓS R et al. Adapting Bobbert-Vlieger model to spectroscopic ellipsometry of gold nanoparticles with bio-organic shells[J]. Biomedical Optics Express, 2017, 8(8): 3538.
[17] ARWIN H. Application of ellipsometry techniques to biological materials[J]. Thin Solid Films, 2011, 519(9): 2589-2592.
[18] ZIMMER A, VEYS-RENAUX D, BROCH L et al. In situ spectroelectrochemical ellipsometry using super continuum white laser: Study of the anodization of magnesium alloy [J]. Journal of Vacuum Science & Technology B, 2019, 37(6): 062911.
[19] ZANGOOIE S, BJORKLUND R, ARWIN H. Water Interaction with Thermally Oxidized Porous Silicon Layers[J]. Journal of The Electrochemical Society, 1997, 144(11): 4027-4035.
[20] KYUNG Y B, LEE S, OH H et al. Determination of the optical functions of various liquids by rotating compensator multichannel spectroscopic ellipsometry[J]. Bulletin of the Korean Chemical Society, 2005, 26(6): 947-951.
[21] OGIEGLO W, VAN DER WERF H, TEMPELMAN K et al. Erratum to ― n-Hexane induced swelling of thin PDMS films under non-equilibrium nanofiltration permeation conditions, resolved by spectroscopic ellipsometry‖ [J. Membr. Sci. 431 (2013), 233-243][J]. Journal of Membrane Science, 2013, 437: 312..
[22] BROCH L, JOHANN L, STEIN N et al. Real time in situ ellipsometric and gravimetric monitoring for electrochemistry experiments[J]. Review of Scientific Instruments, 2007, 78(6).
[23] BISIO F, PRATO M, BARBORINI E et al. Interaction of alkanethiols with nanoporous cluster-assembled Au films[J]. Langmuir, 2011, 27(13): 8371-8376.
[24] 李广立. 氧化亚铜薄膜的制备及其光电性能研究[D]. 西南交通大学, 2016.
[25] 董金矿. 氧化亚铜薄膜的制备及其光催化性能的研究[D]. 安徽建筑大学, 2014.
[26] 张桢. 氧化亚铜薄膜的电化学制备及其光催化和光电性能的研究[D]. 上海交通大学材料科 学与工程学院, 2013.
[27] DISSERTATION M. Cellulose Derivative and Lanthanide Complex Thin Film Cellulose Derivative and Lanthanide Complex Thin Film[J]. 2017.
[28] NIE J, YU X, HU D et al. Preparation and Properties of Cu2O/TiO2 heterojunction Nanocomposite for Rhodamine B Degradation under visible light[J]. ChemistrySelect, 2020, 5(27): 8118-8128.
[29] STRASSER P, GLIECH M, KUEHL S et al. Electrochemical processes on solid shaped nanoparticles with defined facets[J]. Chemical Society Reviews, 2018, 47(3): 715-735.
[30] XU Z, CHEN Y, ZHANG Z et al. Progress of research on underpotential deposition——I. Theory of underpotential deposition[J]. Wuli Huaxue Xuebao/ Acta Physico - Chimica Sinica, 2015, 31(7): 1219-1230.
[31] PANGAROV n. Thermodynamics of electrochemical phase formation and underpotential metal deposition[J]. Electrochimica Acta, 1983, 28(6): 763-775.
[32] KAYASTH S. ELECTRODEPOSITION STUDIES OF RARE EARTHS[J]. Methods in Geochemistry and Geophysics, 1972, 6(C): 5-13.
[33] KONDO T, TAKAKUSAGI S, UOSAKI K. Stability of underpotentially deposited Ag layers on a Au(1 1 1) surface studied by surface X-ray scattering[J]. Electrochemistry Communications, 2009, 11(4): 804-807.
[34] GASPAROTTO L H S, BORISENKO N, BOCCHI N et al. In situ STM investigation of the lithium underpotential deposition on Au(111) in the air- and water-stable ionic liquid 1-butyl-1-methylpyrrolidinium bis(trifluoromethylsulfonyl)amide[J]. Physical Chemistry Chemical Physics, 2009, 11(47): 11140-11145.
[35] SARABIA F J, CLIMENT V, FELIU J M. Underpotential deposition of Nickel on platinum single crystal electrodes[J]. Journal of Electroanalytical Chemistry, 2018, 819(V): 391-400.
[36] BARD A J, FAULKNER L R, SWAIN E et al. Fundamentals and Applications[M]. John Wiley & Sons, Inc, 2001.
[37] SCHWEINER F, MAIN J, FELDMAIER M et al. Impact of the valence band structure of Cu2O on excitonic spectra[J]. Physical Review B, 2016, 93(19): 1-16.
[38] XIONG L, HUANG S, YANG X et al. P-Type and n-type Cu2O semiconductor thin films: Controllable preparation by simple solvothermal method and photoelectrochemical properties[J]. Electrochimica Acta, 2011, 56(6): 2735-2739.
[39] KAZIMIERCZUK T, FRÖHLICH D, SCHEEL S et al. Giant Rydberg excitons in the copper oxide Cu2O[J]. Nature, 2014, 514(7522): 343-347.
[40] RAEBIGER H, LANY S, ZUNGER A. Origins of the p-type nature and cation deficiency in Cu2 O and related materials[J]. Physical Review B - Condensed Matter and Materials Physics, 2007, 76(4): 1-5.
[41] 舒云. Cu2O薄膜的电化学制备及其光电化学性能的研究[D]. 云南大学物理与天文学院,2019.
展示全部 